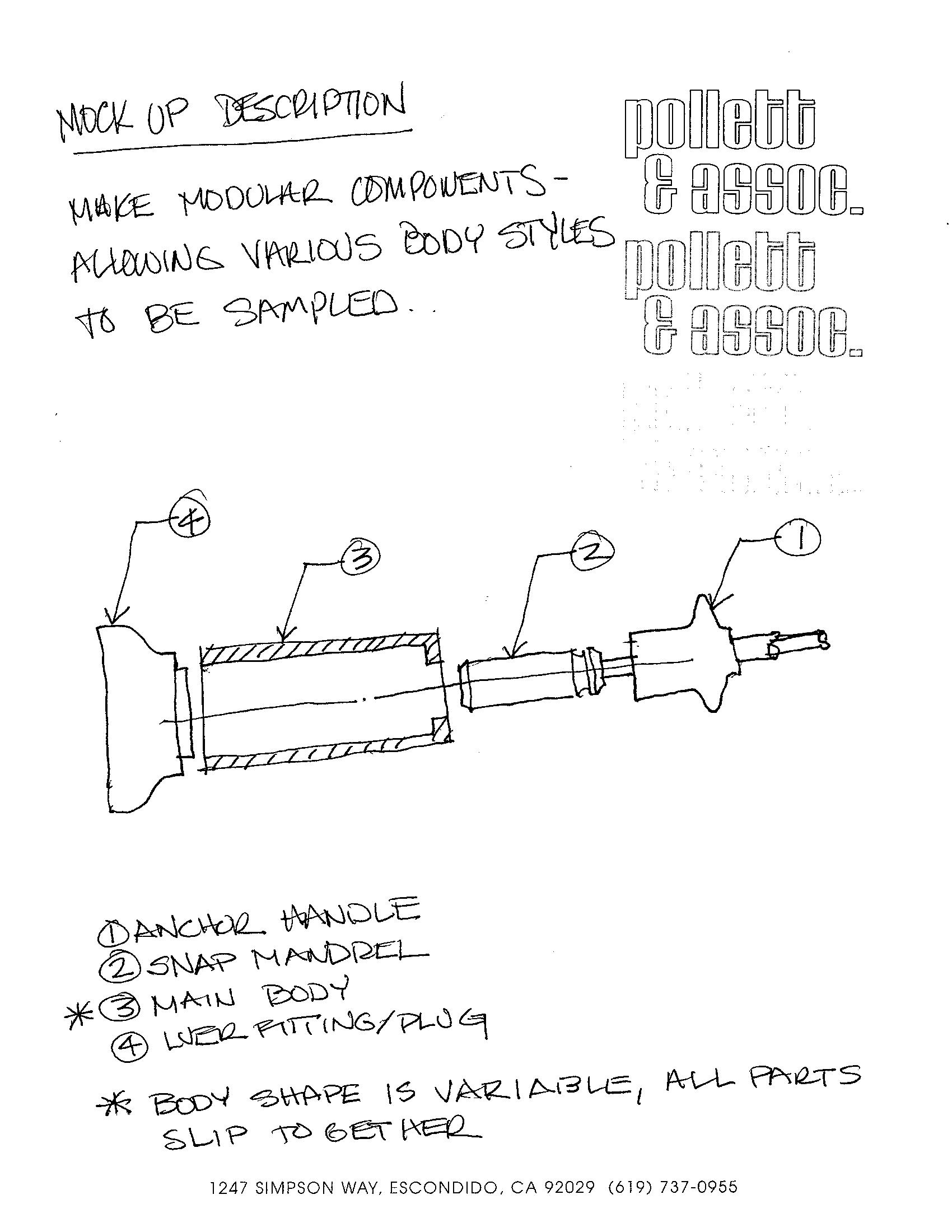
Engineering Conceptualization for Medical Device
More posts from WOODROW POLLETT
View posts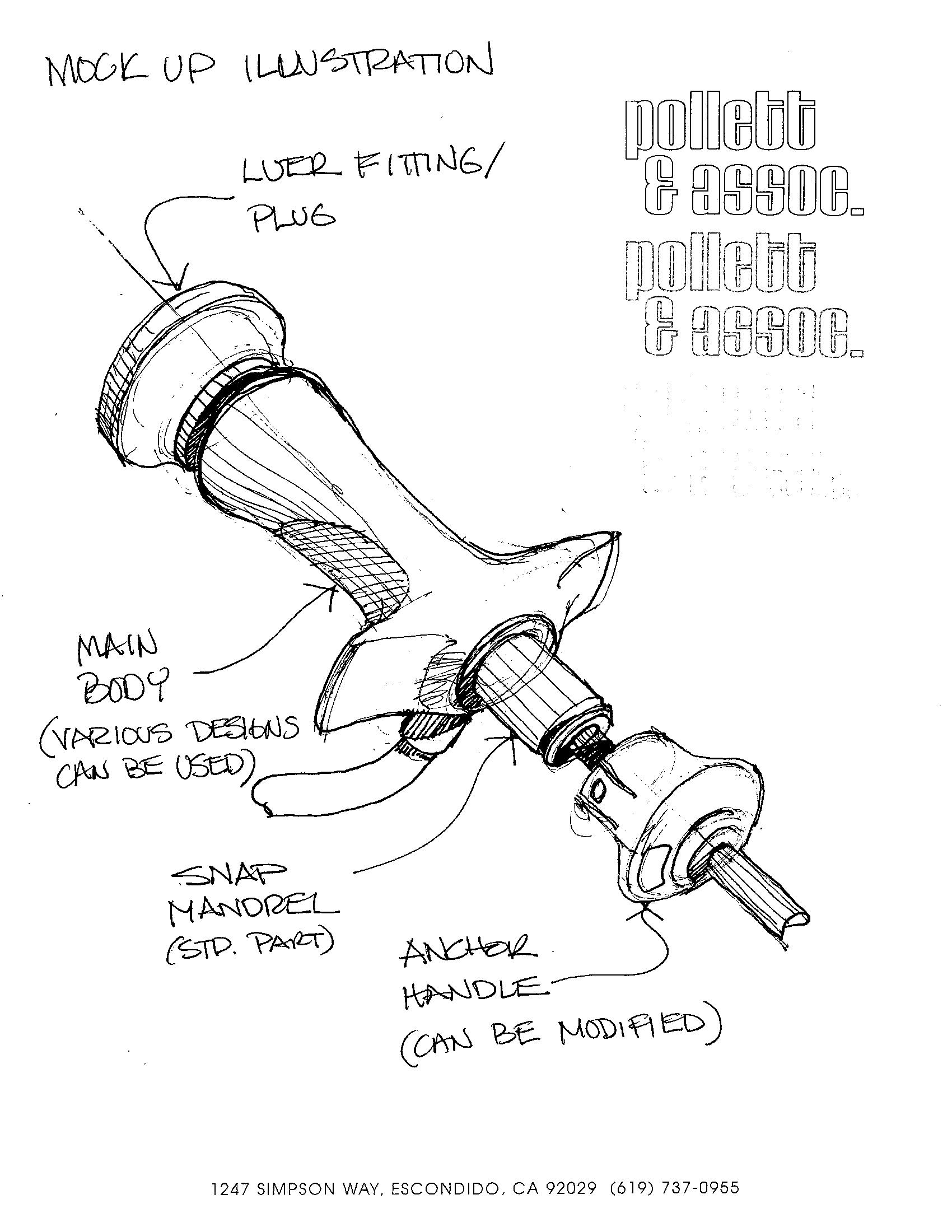


You may be interested in these jobs
-
We are seeking a Quality Engineer to support quality systems and compliance activities for medical device development and manufacturing. · Support and maintain Quality Management System (QMS) in compliance with FDA 21 CFR 820 and ISO 13485. · Participate in design controls , incl ...
North Carolina1 month ago
-
In this role, the Manufacturing Test Engineer will be responsible for developing creative new solutions and improvements to existing designs of test equipment. · ...
Skaneateles2 weeks ago
-
· Job Title: System Engineer · Location: Orlando, FL (Onsite) · Job Description: · Systems Engineering in the medical area is what is needed and experience with these specs: · In depth knowledge performing risk management per ISO 14971 · In depth knowledge in developing and test ...
Orlando, Florida, FL8 hours ago


Comments